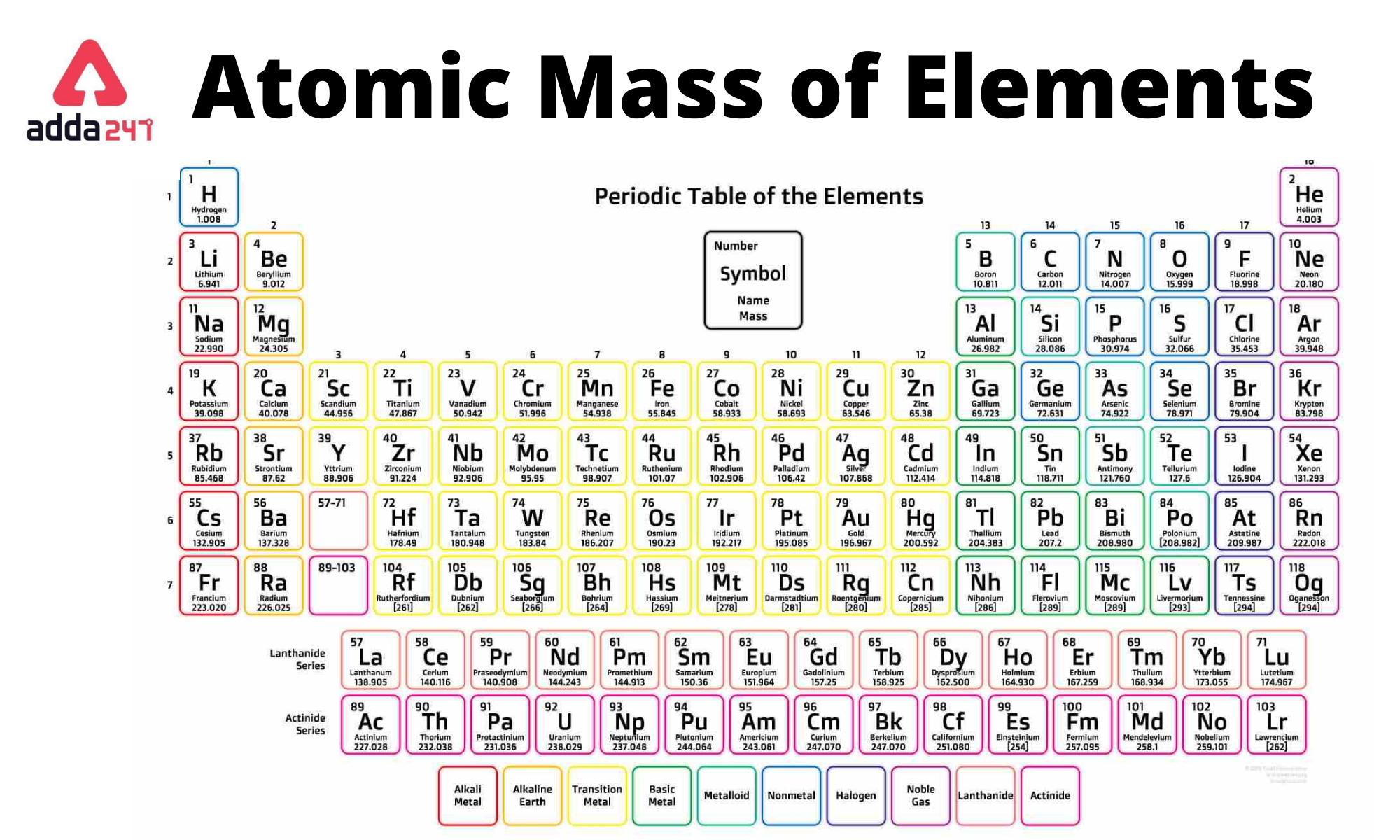
Having a full valence shell of 8 electrons means that the atom is already in its most stable state, and thus, there is no need for the atom to lose or gain electrons. Periodic table with valence electrons is shown in the above image. Noble gases are highly unreactive due to their full outer shell of electrons. 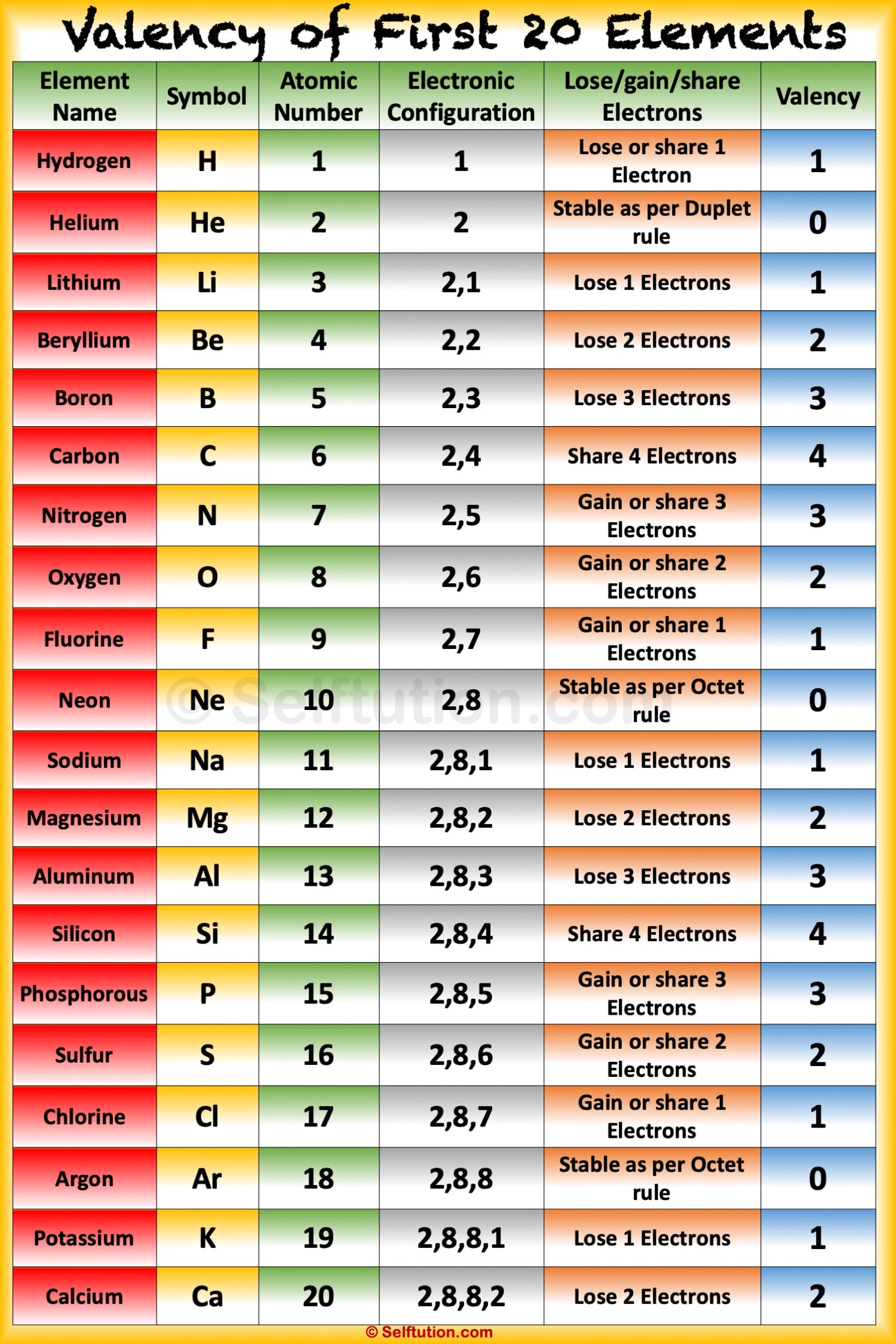
Identify elements that will have the most similar properties to a given element. This group includes helium, neon, argon, krypton, xenon, radon, and the synthetic element oganesson. Explain the relationship between the chemical behavior of families in the periodic table and their valence electrons. I have explained the concept of valence electrons for d block and f block elements in separate. Here is a Periodic Table of elements with everything you need to know. Noble gases are a group of nonmetals in group 18 that are often described as chemically inert - they are colorless, odorless and highly unreactive. Youll get labeled Periodic table with Name, Atomic mass, Electron configuration, Electronegativity, Ionization energy, Electron affinity etc. This was the first element discovered of the group that came to be known as the noble gases. As a result, elements in the same group often display similar properties and reactivity. The elements in each group have the same number of valence electrons. Noble gases were discovered in 1895, when Sir William Ramsay and Lord Rayleigh isolated an unreactive chemical substance that was heavier than nitrogen from the air. The s-, p-, and d-block elements of the periodic table are arranged into 18 numbered columns, or groups. Noble gases are relatively inert and nonreactive due to their full outer shell of electrons. They are all nonmetals, and are found in their standard state as monatomic gases. These are the elements in the 18th column of the periodic table, at the far right. 
Group 18 of the periodic table contains the noble gases. They do not seek to lose or gain electrons, meaning they do not easily react with other elements. They have a full outer shell of electrons, making them already in their most stable state. Noble gases are highly stable because they of their electronic configuration. Noble gases have a full valence shell of 8 electrons. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. How many valence electrons do noble gases have? Design Copyright © 2017 Michael Dayah (). Because they are already in the most stable electronic configuration, they do not easily gain or lose electrons.ģ. Noble gases are unreactive due to their full valence shells. They are chemically inert, colorless, odorless nonmetal elements with a full shell of valence electrons. The noble gases make up group 18 of the periodic table. Elements in the periodic table can be described as metals, metalloids, and nonmetals Elements in the same group typically have similar chemical properties as a result of their similar electronic configuration. All of the elements in a group share the same number of valence electrons: electrons in their outermost shell. Refresher: The periodic table is organized into groups - where each column comprises a group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
